Late Transition Metal Complexes of N-Heterocyclic Carbenes (NHCs) in C–H oxidation.
Small Molecule Activation and C–H B and Oxidation
Late Transition Metal Complexes of N-Heterocyclic Carbenes (NHCs) in C–H oxidation
Activation of naturally abundant small molecules (e.g. O2, N2, CO2, etc.) which are often of high thermodynamic stability is a primary step in the conversion of saturated hydrocarbons to value-added products. This activation process is usually facilitated via a bonding interaction between the molecule and a d-metal ion in a proper environment. In biological systems, the earth-abundant Fe and Mn react with O2 to form highly oxidized metal-oxo species which are reactive intermediates in many oxidation processes. In this project, we are focused on developing NHC-based synthetic systems that can stabilize the high oxidation states of earth-abundant transition metals (Fe, Mn, Co and Cu) (Figure 1). Our studies are geared toward generating structure-function relationships in order to understand the underlining features that lead to formation of these species and how their subsequent reactivity can be modulated.
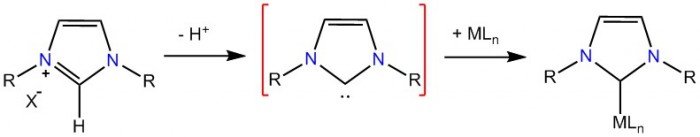
Figure 1.
C–H oxidation
Selective oxidation of C–H bonds in petroleum-based hydrocarbons has been a long-standing challenge in the development of value-added products from cheap and abundant materials (Figure 2). A major part of this difficulty is a result of the high stability of C-H bonds in alkanes which demands harsh reaction conditions (Temperature and oxidizing reagents) to be activated. A number of alkane oxidation catalysts have been developed over the last 60 years including simple metal salts, heme- and non-heme based iron complexes, and polyoxometalates. However, many of the reported systems have shown low conversions and low selectivities due to either over-oxidation of alkane or decomposition of the catalyst under oxidizing conditions. Therefore, there is an indisputable need for catalysts/catalytic systems that can work selectively under ambient conditions to avoid over-oxidation, radical chain reactions, and catalyst decomposition.
TM-NHC Complexes and C–H oxidation
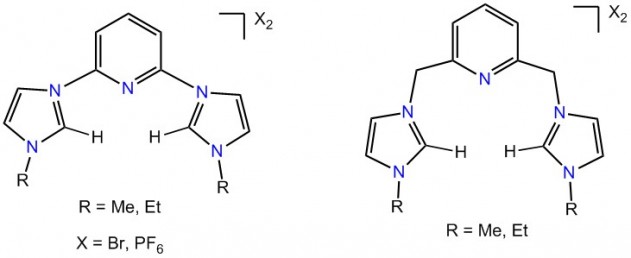
Since the first isolation of a thermally stable carbene and first application of Pd-NHC complexes as catalysts in the Heck coupling reactions, the field of organometallic catalysis has experienced a tremendous growth over the past two decades. The easy synthesis of NHC precursors, high modularity, and strong σ-donor property which allows strong bonding to metals have made them extremely popular ligands in homogeneous catalysis. However, the literature precedents of TM-NHC complexes in oxidation catalysis are restricted to a few precious metals. Complexes of Pd(0) and Pd(II) have been used for stoichiometric oxidation of O2 and CO2 or catalytic oxidation of alcohols and methane. Despite the well-known strong σ-donor and partial π-acceptor properties of NHCs and their potential coordination to soft metals, the high oxidation states of 3d metals are far less developed than their low-valent species. This drawback is due to the relatively soft character of the carbene center in NHCs that favors coordination to soft metals. In this project we work on synthesis of relatively hard NHCs to access the high oxidation states of transition metals. Some of the carbene precursors that have been prepared in the group are shown (Figure 3).
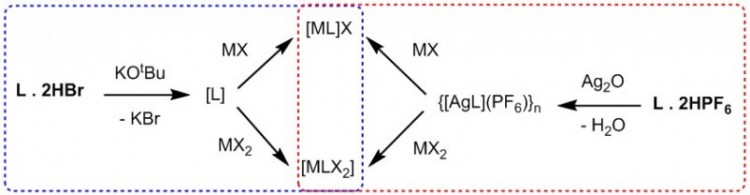
Late transition metal complexes of the NHCs are prepared by either the direct reaction of a metal precursor and free carbene or the transmetallation of a silver-NHC complex with a desired metal salt. As shown in the dotted blue frame of Figure 4, the free carbenes are generated through the reaction of L.2HX and common bases like KOtBu. The metal precursor reacts with the in-situ generated carbene to form the metal-NHC species. In the second method (dotted red frame, Figure 4), a silver-NHC species is generated which undergoes a metathesis reaction with metal precursors to form the TM-NHC complex.
Characterization Techniques
We employ the common characterization techniques such as NMR (1H and 13C), Elemental Analysis, UV-Vis Spectroscopy and X-ray Crystallography to identify our prepared NHC precursors and their transition metal complexes. To investigate their electron-transfer properties and reactivities in the oxidation reactions we use electrochemical techniques such as CV, DPV, and CPE as well as kinetic analysis.
